It was five years ago — April 2, 2013 — that the BRAIN (Brain Research through Advancing Innovative Neurotechnologies®) Initiative went into motion. The goal: to deepen understanding of the inner workings of the human mind and to improve how we treat, prevent, and cure disorders of the brain. Since then, a key player in the project, the National Institutes of Health (NIH), has invested more than $559 million in the research of more than 500 scientists.
The effort also spans other government agencies, including the National Science Foundation, the Defense Advanced Research Projects Agency, and the Intelligence Advanced Research Projects Activity, as well as private foundations, institutions, and companies. Congress continues to show support for the project, appropriating close to $400 million in NIH funding for fiscal year 2018.
While much remains to be discovered, the Initiative has already delivered a number of new tools to study the brain, from new methods for mapping individual neural connections to improved devices for monitoring the active human brain. Some researchers are already testing new technologies to help people with depression, paralysis, and other disorders.
“It’s really had a huge impact on neuroscience,” says Richard Huganir, president of the Society for Neuroscience, who also directs the neuroscience department at the Johns Hopkins University School of Medicine in Baltimore. “Because of the complexity of the brain, we really needed to develop better techniques and tools.”
“The first few years of the BRAIN Initiative have focused on just that, filling the neuroscience toolbox,” says Walter Koroshetz, director of the National Institute of Neurological Disorders and Stroke in Bethesda, Maryland. Moving forward, researchers will continue to develop these tools and employ them to decipher the brain.
A Closer Look
Huganir is particularly excited about new imaging tools enabled by BRAIN Initiative grants. Scientists can now use advanced microscopy techniques to peer into the brains of living lab animals. They can watch as different nerve cells, or neurons, turn on. Or they can see when those cells release neurotransmitters to communicate with each other. “People are imaging when the animal learns something, watching how neurons respond or change their connectivity,” Huganir says. “If you told me 10 years ago that we would be able to do to that, I would say you were crazy.”
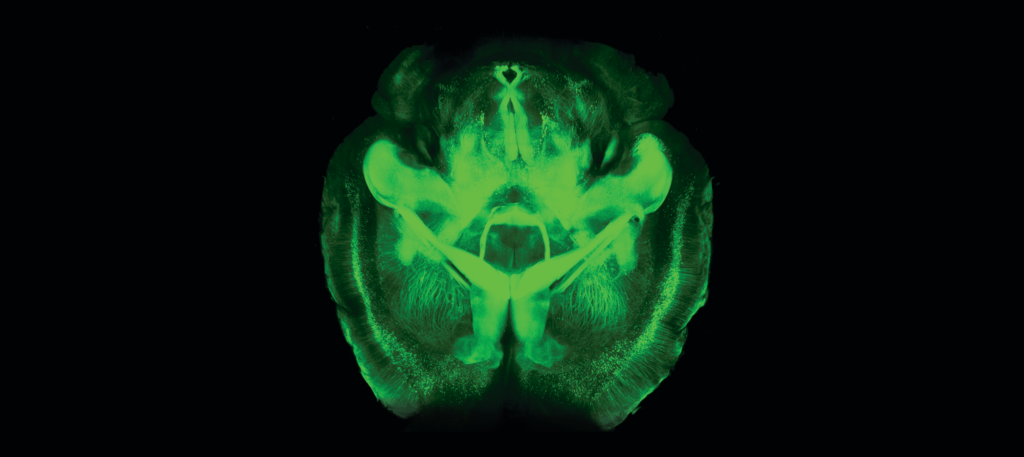
CLARITY immunostaining makes a mouse brain completely transparent. Image courtesy of the Chung lab at MIT.
Other tools help researchers create detailed brain maps in lab animals. Launched in 2017 by the NIH, the BRAIN Initiative Cell Census Network is building a reference ‘atlas’ of all the cell types in the brain — including not only neurons, but the many other cells that support them.
In addition to cataloguing the key components of the brain, researchers are tracing how neurons fit together. The end result, called the connectome, is a network mapping all the neural circuits in a section of the brain. The brain’s extreme density makes this a challenging task, like mapping a tangle of beaded necklaces. But novel microscopy and other techniques provide unprecedented access. For example, researchers have used BRAIN funding to refine a tool called CLARITY, which renders an entire preserved brain transparent. That makes it possible to observe how different types of neurons are connected across long distances in the brain.
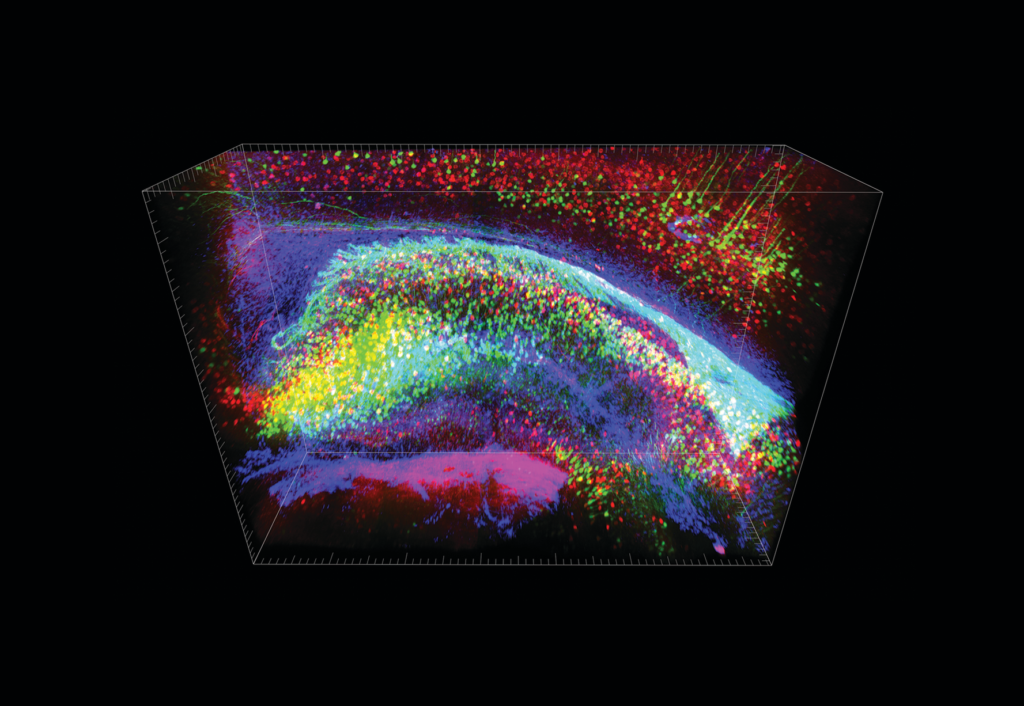
CLARITY enables better visualization of neurons. Image courtesy of the Chung lab at MIT.
“That’s important because many psychological and neurological disorders cause miscommunication in the brain’s circuitry, leading to disabling symptoms,” Koroshetz says. “Throughout history, we have not been able to see the abnormal circuit activity, so trying to fix it has been very hard,” he says.
Into the Human Mind
To that end, BRAIN-funded scientists are also developing and updating ways to fix circuitry gone awry. One approach is deep brain stimulation (DBS), which involves implanting a tiny electrode into a part of the brain that’s misfiring and delivering a steady current. DBS has been approved since 1997 to quell the shakiness experienced by people with Parkinson’s disease and another condition, called essential tremor.
“It’s amazing to see and drives the questions, what if we had precise tools to manipulate brain circuits to quell chronic pain, depression, seizures, and other disorders,” says Koroshetz.
At the University of Florida in Gainesville, biomedical engineer Aysegul Gunduz hopes to make DBS technology smarter. She likens today’s version to an air conditioner set to fan mode — it provides the same steady breeze, regardless of the temperature. Similarly, DBS currently involves setting the system to a single, constant stimulation level.
But AC works better when there’s a thermostat, so it can adjust cooling as needed. So might DBS, Gunduz says. For example, some people with essential tremor only shake during purposeful actions — reaching for a cup of water, for example. But the stimulation can also cause annoying side effects, such as stuttering. In fact, some people turn off their DBS device when they want to speak. What if, instead, the DBS device could turn itself on only when needed?
Gunduz’s lab is testing new DBS devices, developed by Medtronic of Minneapolis, that can simultaneously deliver stimulation and record what the brain is doing. In a clinical trial funded by the BRAIN Initiative, Gunduz and colleagues will implant the sensor-stimulators in growing numbers of people with essential tremor. The data they collect will help scientists better understand the neural signals that control tremors. The hope is that future stimulators could identify when a person is about to move and turn on the current only at those times.
Helen Mayberg, director of the Center for Advanced Circuit Therapeutics at the Icahn School of Medicine of Mount Sinai in New York City, works with the same sensors, but she’s targeting depression. Over years of research, she’s found that DBS of a particular part of the brain, called area 25, can help people with treatment-resistant depression. She’s treated dozens of people in this way.
Mayberg and collaborators at Emory University in Atlanta have BRAIN Initiative funding to test Medtronic’s sensor-stimulators. “This is going to teach us quite a bit about how area 25 participates in depression, and depression recovery,” says Mayberg.
Both Mayberg and Gunduz’s projects are enabled by the BRAIN Initiative’s Public-Private Partnership Program, which links clinical researchers with companies developing new neurotechnologies. By fostering collaboration between science and industry, the program ensures that new clinical studies can leverage existing data from device manufacturers, ultimately speeding research.
Mind in Motion
Other efforts are focused on helping people who are paralyzed or ‘locked-in,’ including those with spinal injuries or ALS (Lou Gehrig’s disease). The BrainGate project is testing a brain-machine interface designed to help people use neural activity to control a computer or robotic arm. Researchers won BRAIN funding in 2015 to support expanded clinical testing of the device, which they’ve been developing for more than a decade.
The implant, about the size of a pinky fingernail, houses an array of 100 hairlike needles that track electrical activity of 100 to 200 nearby neurons. Surgeons implant this array into the part of the brain that controls movement of the right arm. A computer chip then converts neural activity from the sensor into the motion of a cursor on a tablet or computer screen. It only takes about five minutes to train someone to use the system.
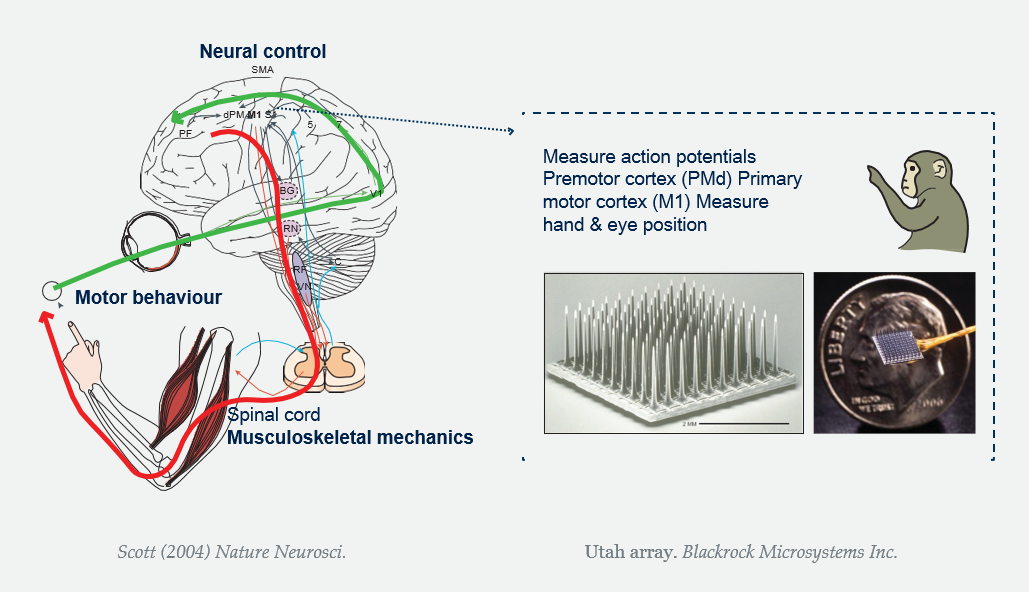
Scott (2004) Nature Neurosci. Utah array. Blackrock Microsystems Inc.
“Just by thinking about moving their right arm, they can move a computer cursor over a keyboard and type,” says Krishna Shenoy, a neuroscientist at Stanford University in California. “It’s now possible to type about eight words per minute without errors.” And that’s without autocorrect or word completion, he adds.
For now, the scientists are most concerned with the safety of the implants. However, they’ll also be checking how well the implants work, in the hopes of planning a larger trial in the future.
Looking Forward
The BRAIN Initiative is set to run through at least 2026. Over the next eight years, scientists expect that BRAIN-funded technologies will enable novel discoveries about how the brain works. These insights will eventually translate into treatments for people with a variety of neurological conditions, predicts Joshua Gordon, director of the National Institute of Mental Health in Bethesda.
“Neurologic and psychiatric disease, collectively, are the number one cause of disability worldwide,” he points out. “They have a huge, huge burden to society, as well as the families.” Anything that alleviates that burden will help not only the people receiving new treatments, but the world economy too, he says.
The brain remains complex — but thanks to the BRAIN Initiative, it’s more and more comprehensible.

